Menu
- Molecular Hydrogen Overview:
-
Hydrogen Chemistry:
- H, H2 H+, H-, Carbohydrates and Isotopes:
- Oxidation, Reduction and Redox:
- ROS, Oxidative Stress and Antioxidants:
- Concentration, Saturation & Half-Life:
- pH and ORP:
- Alkaline Water versus Hydrogen-Rich Water:

Molecular Hydrogen Overview
Molecular hydrogen (H2) is an amazing supplement, which has gained worldwide attention for its therapeutic potential. Although still in its infancy, research on molecular hydrogen has accelerated ever since a 2007 clinical study demonstrated that administration of hydrogen gas exerted therapeutic biological effects.
- Therapeutic effects now attributed to H2 include:
- Anti-aging
- Reduces excessive oxidative stress
- Supports anti-inflammation
- Supports healthy detoxification
- Supports healthy, hydrated and elastic skin
- Supports the protection of DNA and RNA
- Increases energy levels
- Improves efficiency of ATP production
- Stimulates mitochondrial biogenesis
- Improves exercise and sports performance and recovery
- Lowers blood lactate levels to help delay fatigue
- Increases VO2 max
- H2 has numerous properties that enable it to exert these effects:
- H2 is VERY bioavailable:
- The high degree of molecular hydrogen's bioavailability is due to its:
-
very small size and weight.
- (the smallest and lightest molecule in the universe)
- neutral charge
- non polar structure
- Molecular hydrogen's high degree of bioavailability allows it to:
- Easily diffuse through out the body
-
Rapidly penetrate biomembranes
- cell membranes, blood-brain barrier, placental barrier, and testis barrier
-
Reach subcellular compartments
- mitochondria, nucleus, etc.
- H2 acts as a very selective antioxidant.
- Can DIRECTLY neutralize free radicals
-
reacts primarily with the hydroxyl radical
- (the most cytotoxic free radical in the body)
- When H2 neutralizes the hydroxyl radical, it produces H2O as a result.
- Through cell modulation, H2 can INDIRECTLY reduce excessive oxidative stress.
- Stimulates metabolic pathways by signal transduction:
- Can activate numerous transcription factors which lead to altered gene expressions.
- H2 can activate the Nrf2-Keap1 pathway.
- which produces over 300 cytoprotective antioxidants and proteins
- This activation only happens when there is excessive oxidative stress.
- H2 can down-regulate the NADPH oxidase system.
- This only happens when there is excessive oxidative stress.
- H2 can be administered numerous ways including:
- inhalation
- ingestion of solubilized (dissolved) hydrogen-rich solutions (e.g. water, flavored beverages, etc.)
- hydrogen-rich hemodialysis solution
- intravenous injection of hydrogen-rich saline
- topical administration of hydrogen-rich media (e.g. bath, shower, and creams)
- hyperbaric treatment
- ingestion of hydrogen-producing material upon reaction with gastric acid
- ingestion of non-digestible carbohydrates as prebiotic to hydrogen-producing intestinal bacteria
- rectal insufflation and other methods.
- The simplest, most convenient, safest and most effective method is by drinking HRW made by dissolving H2 forming effervescent tablets into water.
- H2 Max provides the maximum amount of H2
Hydrogen Chemistry
In order to better understand the therapeutic effects of molecular hydrogen, we must first understand some basics about the chemistry of hydrogen.
H2 is a colorless, odorless and flammable gas that was previous thought to be unreactive and physiologically inert in mammalian cells, but is now known to be a reducing agent (an antioxidant) in addition to having some other very unique biological, health supporting properties.
The incorrect thinking about H2 in the past was based on the fact that H2 is a neutral (uncharged), non polar, small molecule, which is not big enough to bind to any protein receptors or protein channels. Although, it is these exact properties that make H2 easy to reach cells . . . it was erroneously postulated, that once inside cells, H2 would be incapable of exhibiting any biological activity.
However in 2005, ground breaking research conducted by Dr. Shigeo Ohta et al (out of the Nippon Medical School, Japan) revealed that H2 has antioxidant properties, moreover it has selective antioxdant properties. Dr Ohta's study Hydrogen acts as a therapeutic antioxidant by selectively reducing cytotoxic oxygen radicals (published in "Nature Medicine" June 2007) was single handedly responsible for giving birth to the hydrogen research revolution.
Since that landmark discovery, over 1500 studies (using diverse human and animal models) have consistently shown that hydrogen is capable of producing some very unique and significant biological effects, which support health.
Let's dig into the chemistry of hydrogen.
Hydrogen can exists in several forms:
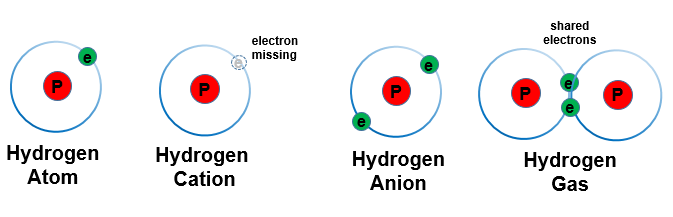
- Atomic Hydrogen (H)
- The first (and lightest) element in the periodic table of elements.
- It is not stable and tends to bind to another H atom to form H2.
-
Each hydrogen atom has:
- one proton (a positively charged particle)
- one electron (a negatively charged particle).
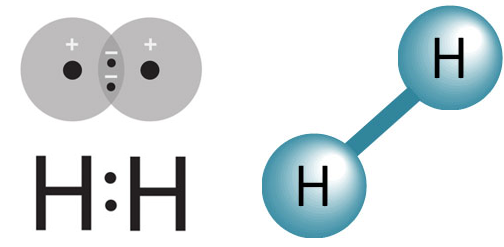
- Molecular Hydrogen (Di-hydrogen gas) (H2)
- the smallest and most abundant molecule in the universe.
- Each hydrogen molecule has:
- two protons
- two electrons
- Hydrogen bound to other substrates, for example:
- water (H2O)
- carbohydrates, such as glucose (C6H12O6)
- Ionic Hydrogen
- Hydron (a positively charged hydrogen ion) (H+)
- a hydrogen atom that has lost its electron (a proton)
- responsible for making pH (potential hydrogen)
- negative logarithm of H+ concentration
- Hydride (a negatively charged hydrogen ion) (H-)
- a hydrogen atom with two electrons
- not stable and does not exist in nature
Its worth mentioning that atomic hydrogen also has two isotopes (heavy atoms).
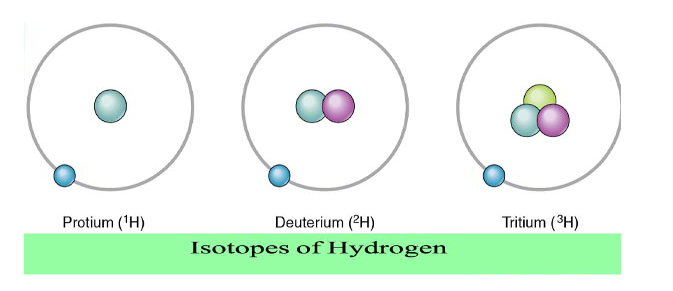
- Deuterium
- can have some negatve biological effects
- in addition to one electron and one proton, it also has
- one neutron
- Tritium
- radioactive so it has very negative biological effects
- in addition to one electron and one proton, it also has
- two neutrons
Hydrogen is the only element, whose isotopes are distinguished from each other with different common names. Because the normal hydrogen atom, technically belongs to the set of hydrogen isotopes, it also has an isotope style name . . . protium. Likewise, hydrogen gas can also be referred to as di-protium.
Its important to note that when we speak of hydrogen ingestion and hydrogen research, we are referring strictly to molecular hydrogen (H2).
Sometimes people get confused and think we are talking about alkaline water, because they associate pH with hydrogen, which is totally unrelated to the hydrogen properties we are discussing here.
In fact, the latest research indicates that most of the health benefits attributed to drinking alkaline water, are related to its H2 content (a result of the ionization process) NOT its alkalinity.
Studies have demonstrated that drinking alkaline water (not containing H2) provides no more health benefits than drinking pure distilled water, while drinking hydrogen-rich water (not alkaline) does provide the benefits.
I want to emphasize the point, that hydrogen-rich water does not change the pH or alter the structure of water. It is simply water with molecular hydrogen dissolved in it. The water acts as a carrier for the hydrogen. And because the hydrogen does not react or combine with the water molecules, it dissipates rather quickly, which is why its important to drink hydrogen-rich water, soon after it is made, before the H2 can escape.
So, whether it be inhaled, drank as infused water, applied topically etc., we are referring to molecular hydrogen.
Read LessOxidation, Reduction and Redox
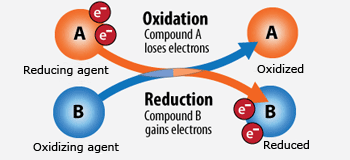
Oxidation is loss of electrons. Reduction is gain of electrons. (Easily remembered by the mnemonic device OIL RIG).
Redox is a chemical reaction in which oxidation and reduction occur simultaneously. In other words . . . a chemical reaction where electrons are exchanged between two reactants:
1) The reducing agent (aka antioxidant) causes reduction by giving electrons to the oxidizing agent (reducing it) and in doing so becomes oxidized itself (its oxidative state increases).
2) The oxidizing agent (aka oxidant) causes oxidation by accepting electrons from the reducing agent (oxidizing it) and in doing so becomes reduced itself (its oxidative state decreases).
The simplest way to think of this is that:
- the oxidizing agent is the substance that gets reduced,
- while the reducing agent is the substance that gets oxidized.
Examples of Redox reactions include: water treatment, photosynthesis, corrosion, combustion and explosions.
- Reducing agents include H2 and most metals
- Oxidizing agents include H2 and nonmetals such as: like F, Cl and O.
- As you can see, H2 can act as either a reducing agent (antioxidant) or oxidizing agent (oxidant)
Free Radicals, Oxidative Stress and Antioixidants
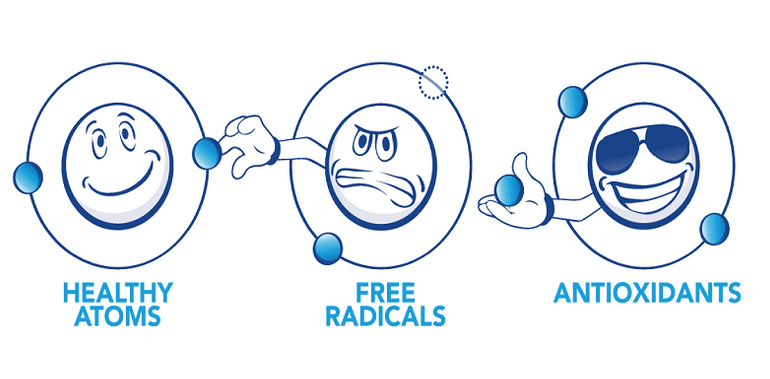
Metabolism is an essential process whereby broken down food is converted to energy. Oxygen is consumed in this process and CO2 is a waste by-product (among others) that is produced at the same time. This metabolic process of "burning" fuel to produce energy involves the harvesting of electrons from other molecules. A side effect of this process is the production of a Reactive Oxygen Species (ROS) which are special types of free radicals.
Molecules are stable when their electrons are in pairs, with each electron spinning in the opposite direction in the same shell, but free radicals are extremely unstable because of the missing electrons that were harvested from them during the metabolic process. They have a strong affinity for other electrons in order to re-establish their pair and become stable again, so they tend to scavenge other atoms, molecules or cells in order to steal the electrons they need to complete their outer shell.
Once free radicals are formed, they create a chain reaction that destabilizes other molecules, causing damage to cells, proteins, and DNA. This chain reaction (known as the cascading effect) may also alter cell membranes, affecting what molecules can enter and exit cells, and may even change the structure of certain lipids (fats) which may make them more likely to become trapped in an artery.
Free radicals have been associated with premature agin and many human diseases including cancer, atherosclerosis, Alzheimer's disease, Parkinson's disease, and more.
Aside from the normal metabolic process, other factors contributing to the generation of free radicals include: drinking alcoholic beverages, smoking tobacco, taking some medications, as well exposure to pesticides and air pollutants.
Free radicals are also generated during exercise. The good news is that regular exercise builds up antioxidant defenses. However, out of shape and infrequent exercisers who partake in intense physical activity may suffer from the cell damaging effects of free radicals.
The damage caused to cels and tissues by free radicals is known as oxidative stress. Oxidative stress plays a role is many chronic conditions, such as macular degeneration, cardiovascular disease, emphysema, certain cancers, arthritis, lupus, and more
Antioxidants are molecules that fight free radicals in the body. We have natural antioxidant defenses - pathways that produce antioxidants to fight the damage from the free radicals. We also need to get antioxidants from food, which are mostly found in plant-based, whole foods. In recent years, antioxidant supplements have been more and more popular.
Many people might think that more antioxidants are better. However, high doses of supplemental antioxidants have actually been connected to INCREASED disease risk. The problem is that supplemental antioxidants can soak up free radicals indiscriminately attacking free radicals wherever they find them. But not ALL free radicals are bad. And in fact, some key free radicals, in balanced amounts, are actually essential for healthy cells and healthy bodies.
Concentration, Solubility, Saturation and Half-Life
The concentration of any given gas (a solute) in any given liquid (a solvent) is typically specified in molarity (moles/millter (M) or millimoles/L mM), parts per million (ppm), parts per billion (ppb) or miligrams per liter (mg/L). Since 1 liter of water happens to weigh 1 million milligrams, 1 ppm is equal to 1 mg/L (for dilute concentrations) so the terms are often used interchangeably.
- The concentration of a gas that can be reached in water depends on several factors:
- temperature of the water
- pressure of the gas
- solubility of the gas
- determined by the intrinsic chemical/physical properties of the gas
- size
- charge
- polarity
- Each gas has a different solubility constant.
- known as "gas constants" or "Henry's constants" (KH)
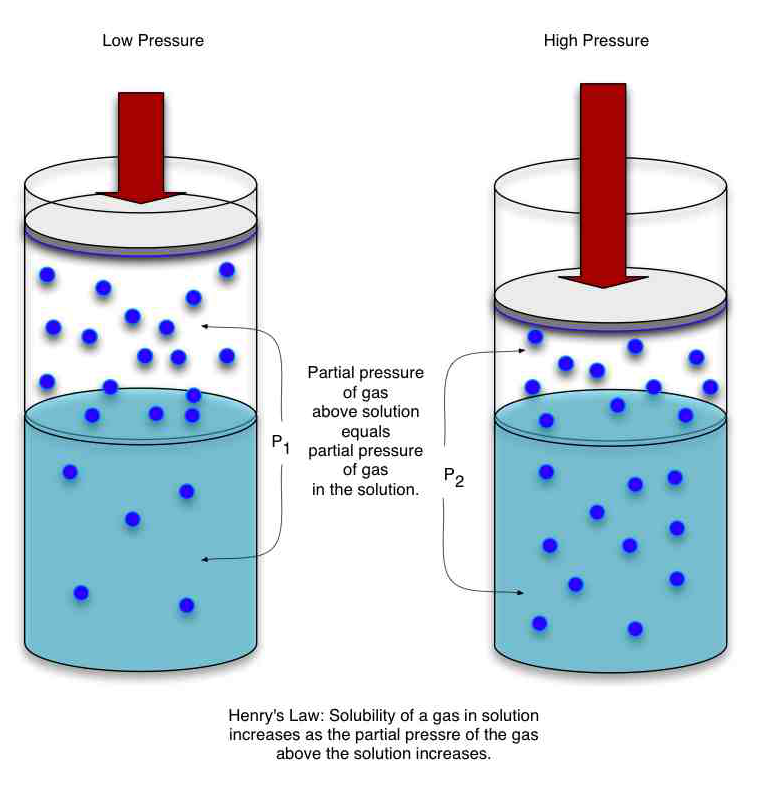
- Henry's law states that the concentration of any gas in water is directly proportional to the partial pressure of that gas above the water.
- "C" represents the concentration of the dissolved gas.
- "KH" represents "Henry's constants" for that gas.
- "P" represents the partial pressure of that gas (above the solution).
- if the pressure of that gas increases, the amount of that gas dissolved in the water also increases.
- This is how carbonated beverages are produced;
- the pressure of CO2 is increased
- resulting in more CO2 being dissolved in the beverage
- Saturation of a gas in water is defined as the concentration obtained, when the pressure of the gas above the solution is at equilibrium with the pressure of the gas in the solution.
- In the case of H2, the saturation concentration in water (at a partial pressure of 1 atm) is 1.6 mg/L.
- A saturated solution such as this can be produced by infusing H2 into water, using an infusion stone that creates ultra small H2 bubbles.
- According to Henry's Law, that saturation concentration can be increased by increasing the pressure of H2 (above the water).
- Just as with the production of carbonated beverages (in the example above)
- Hydrogen Rich Water (HRW) can be produced by dissolving effervescent magnesium tablets into water.
- The resulting H2 concentration using this method can far exceed that of a saturated solution and is thus known as a super saturated gaseous solution.
- Many molecular hydrogen clinical studies have been conducted using H2 dissolved in water.
- typically using concentrations ranging from 0.5 mg/L to 1.6+ mg/L.
- Most of the recent studies use HRW with concentrations near 8 mg/L.
- It's very important to understand that as useful as concentration is to know, it only provides density information (the ratio of the solute's mass to the water's mass), without specifying the water's total volume.
- We often need to know the actual amount of solute dissolved in the water (the dosage).
- I refer to this as the "payload".
- The payload is calculated by multiplying the concentration by the actual volume of water.
- In order to get therapeutic effects from drinking hydrogen water, attention must be paid to both:
- the concentration and the payload.
- The concentration needs to be at least 2 ppm.
- The payload needs to be at least .5 mg.
- So for example: with a concentration of 2 ppm, 250 ml must be drank to provide a dose of .5mg.
- The International Hydrogen Standards Association (IHSA) recommends that the payload should be met by ingesting a maximum volume of 1 L of water.
- Those who desire higher payloads (to match doses of the latest clinical studies) should opt for higher concentrations (in order to not exceed 1 L of water).
- It's worth noting that no toxic effects from higher H2 payloads have been discovered, and that the 1 L limitation is based on effectiveness, not safety concerns.
- The half-life of a gas in solution is defined as the time it takes for half of the gas concentration to dissipate.
- Just as an opened can of soda pop, will get "flat" when exposed to normal atmospheric pressure, so will both saturated hydrogen water and HRW.
- The concentration of H2 will decrease until it is at equilibrium with the partial pressure of H2 in the atmosphere (resulting in a concentration of 8.67 x 10-7 mg/L).
- The rate of H2 dissipation from the water is directly effected by temperature, agitation, and surface area.
- A 500 mL open container of dissolved H2 in water has a half-life of about two hours.
- if left out in the open with no turbulence at room temperature with an initial H2 concentration of 1.6 mg/L, the concentration would likely be around 0.8 mg/L after two hours. However, the dissipation rate is not exactly linear.
- H2 has the highest effusion rate of all gases.
- Due to its small molecular size, H2 can diffuse through all plastic containers.
-
C=P/KH
-
Mg + 2H2O = Mg(OH)2 + H2
pH and ORP
pH and ORP are two important analytical metrics performed on solutions. They are very much related to each other and are very often misunderstood, particularly in relationship to hydrogen. So lets begin by defining each:
pH :
- pH stands for "potential of hydrogen" or "power of hydrogen".
- Its conventionally defined as the negative logarithmic measurement of hydron (H+) concentration in solution.
- pH more precisely relates to the concentration of the hydronium ion (H3O+), because:
- H+ ions are very unstable and don't actually exist in water.
- H+ readily combines to the negatively charged oxygen (of H2O) to form H3O+ and OH-.
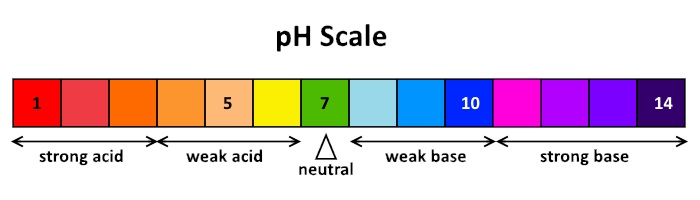
- The pH scale ranges from 1 to 14 (with 7 being neutral).
- The greater the H3O+ concentration . . .
- the more acidic the solution
- indicated by a lower pH
- The lesser the H3O+ concentration . . .
- the more alkaline the solution
- indicated by a higher pH
- Because the scale is logarithmic . . .
- An increase of the pH by 1 unit . . .
- represents a 10 fold decrease in the H3O+ concentration
- An Increase of the pH by 3 units . . .
- represents a 1,000 fold decrease in the H3O+ concentration
- Pure water (at 25° C) is neutral, because:
- The concentration of H3O+ and OH- are equal . . .
- each having a concentration of 10-7 moles/liter
- Because pH is a negative log . . .
- this 10-7 H3O+ concentration represents a pH of 7 (indicating a neutral solution).
- Many people incorrectly think that adding hydrogen gas (H2) to water will change the pH of the water.
- (H2) is a neutral molecule and when dissolved in water, has no influence on the water's pH.
- Methods of producing hydrogen water such as bubbling or infusing H2 into H2O . . .
- do not result in any structural changes to the water (no reactions or ionization)
- the water simply acts as a carrier and thus its pH does not change.
- Alkaline ionizers do raise the pH of the water, but not as a direct result of adding H2.
- In order for these ionizers to produce H2, they must consume the H+ ions in the water, thus making the water more alkaline as a result.
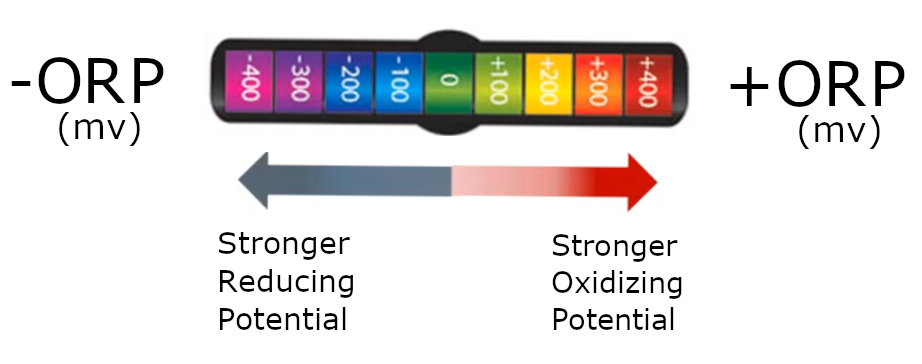
ORP :
- ORP stands for "oxidation-reduction potential" (aka "redox potential")
- ORP is a measurement of water's tendency to act as either:
- a reducing agent (electron donor)
- indicated by a negative ORP
- An oxidizing agent (electron acceptor)
- indicated by a possitive ORP
- The ORP measurement is performed with an ORP meter, by inserting its probe into the water being tested.
- The ORP of H2 water will typically measure some negative value (-100 to -750 mV, depending on pH).
- Its important to understand that the ORP measurement only indicates . . .
- the water's potential to act (as a reducing or oxidizing agent).
- It does not guarantee that any particular oxidation/reduction reaction will actually occur.
- It does not indicate the strength or speed of any potential oxidation/reduction reaction.
- Other factors such as kinetics and thermo dynamics will determine if the potential will ultimately be realized or not.
- Although a negative ORP measurement indicates the presence of a reducing agent . . .
- An ORP meter does not "measure" the concentration of dissolved H2 in the water.
- It measures the relative contributions to the redox potential from two species of hydrogen in the water.
- hydrogen gas (H2)
- hydrogen ions (H+)
- Together, these two species are called a "redox couple".
- Both of the "redox couple's" hydrogen species contribute to the final ORP reading . . .
- producing a small corresponding voltage potential at the probe's electrode
- based upon their relative concentrations and number of electrons
- The meter amplifies and compares the voltage measurement against an internal reference.
- It displays a digital reading (usually in "millivolts").
- The bottom term of the "redox couple" (H+) relates to pH . . .
- This is why the ORP measurement is influenced by the pH of the water being tested.
- While the ORP reading is extremely sensitive to changes in pH . . .
- it is relatively "insensitive" to changes in dissolved H2.
- Even small variations in the water's pH can result in large changes in the ORP reading
- masking the relatively small contribution made to the ORP by the dissolved H2.
- Over a range of 3 pH units . . .
- the ORP will change 178 mV.
- Over a typical range in H2 concentration (0.5 to 2 mg/L) . . .
- the ORP will only change by a total of 18 mV.
- Over their respective ranges . . .
- the pH contributes approximately 90% to the ORP reading
- H2 contributes only about 10%
- This is why, although a negative ORP value may indicate the presence of dissolved H2 . . .
- it cannot be used to quantize its concentration.
- Its important to keep this in mind when comparing hydrogen water made by different methods:
H2/H+
- Alkaline ionizers
- This technology has been around for many decades.
- It uses electrolysis to produce:
-
H2 and Alkaline water
- at the cathode (the negative electrode)
- H2 is produced by reducing (adding electrons to) H+ ions
- Because H+ ions are the acid component in water, their consumption (in this reaction) elevates the pH of the drinking water into the alkaline range (above pH 7) as the hydroxide level (OH-) rises.
2H+ + 2e- = H2
-
O2, CL2 and Acidic water
- at the anode (the positive electrode)
- The anode and cathode are separated by a membrane, which allows for the separation of alkaline and acidic water
- Although many have reported benefits from drinking this alkaline water, studies conducted between 2007-2010 have confirmed that the therapeutic effecs of drinking alkaline water are actually attributed to the hydrogen gas NOT the alkalinity.
- Alkaline water ionizers were optimized and designed to produce alkaline water, not hydrogen gas.
- Most alkaline water ionizers produce insufficient concentrations of dissolved H2 (less than 0.01 ppm) to be therapeutic.
- Some alkaline water ionizers can produce adequate hydrogen gas levels, but due to their production of alkaline water, the electrodes tend to scale up quickly, which prevents the hydrogen gas from dissolving into the water.
- Neutral-pH H2 generators
- There are a number of ways that these types of machines can be designed, but the goal is to produce a high concentration of hydrogen gas without altering the pH.
- Although these machines use electrolysis (just as Alkaline ionizers do), they do not act on the drinking water itself, rather they deploy electrolysis in small isolated chambers (cells) and then infuse the resulting produced H2 into the drinking water, using a special device called a "dissolver", capable of reaching H2 concentrations up to 1.6 ppm,
- Some of the these machines deploy the use of a special membrane in the electrolysis chambers optimized for hydrogen gas production known as a Proton Exchange Membrane (PEM). Machines using this technology can obtain H2 concentrations between 0.8 to 1.5 ppm.
- Because Neutral-pH H2 generators do not change the pH, scaling and subsequent cessation of a high hydrogen concentration is less of a concern than with alkaline ionizers.
- Hydrogen water from a neutral-pH hydrogen water device with its much lower average pH will measure a lower-magnitude negative ORP reading than alkaline water, even if it contains twice as much dissolved H2!
- Dissolving effervescent magnesisum tablets into water
- xxxxxxxxx
Hydrogen and Health

Our bodies contain 62% hydrogen. It is required for every biochemical reaction that takes place in every cell of our bodies, so hydrogen is obviously very important to our health. Hydrogen has numerous properties that contribute to its importance.
Hydrogen is VERY Bioavailable
Because of its small size (the smallest molecule in the universe), its neutral charge and its non polar structure, H2 is able to enter cells without having to travel through any protein transport channels. It can simply follow its concentration gradient and pass through all cell membranes (including the blood-brain barrier) by passive diffusion.
For these reasons, H2 has very strong bioavailability, which allows it to quickly enter the blood stream and easily enter all the cells in the body, as well as their sub cellular compartments, most notably the mitochondria and nucleus.
H2 has some other very special and unique properties that contribute to the major role it plays in supporting optimal health, but none of these properties are more fundamental than its superb bioavailability, for without this property, all of its many other qualities would be far less impactful.
Hydrogen Acts as a Selective Antioxidant.
Metabolism is the essential process whereby the mitochondria in our cells, utilize broken down food and combine it with oxygen to produce energy. Paradoxically, one of the by-products of metabolism is free radicals, which steal electrons from healthy cells. This oxidative stress damages our cells and tissues thus aging our bodies.
Our bodies combat oxidative stress through the production and use of antioxidants.
Hydrogen is a very unique antioxidant because its not only very bioavailable, but it is also highly selective and non-cascading.
- Unlike all other antioxidants, when hydrogen donates electrons to neutralize free radicals, it does not become a free radical itself. In other words it does not contribute to the oxidative cascading effect.
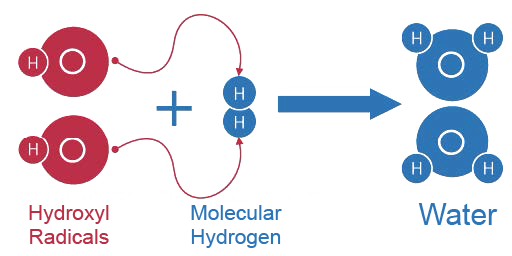
- Hydrogen is selective in that it only reduces toxic free radicals like the hydroxyl radical (the most cytotoxic radical) and in doing so, instead of becoming a free radical itself, it combines with the hydroxy radical to form water.
- Hydrogen also neutralizes peroxynitrite radicals (also very toxic, though to a lesser degree than hydroxy radicals), yet, it does NOT react with any of the other biologically important oxygen radicals (e.g. superoxide, nitric oxide, hydrogen peroxide etc.).
- Our bodies actually need some free radicals, such as the oxides of nitrogen, which are needed to help blood vessels dilate and constrict properly. so taking too many antioxidants can be harmful.
- Athletes taking chronic high levels of antioxidants, can negate the benefits of exercise training, by potentially neutralizing the beneficial signaling molecules produced by exercise.
- The bottom line is that it is important to have a good balance of oxidation and reduction in our bodies and H2 helps reach this balance, bringing everything back to homeostasis.
The ability of H2 to selectivity reduce the toxic radicals, while not neutralizing the ones we need, is a very important feature of H2.
- In order to understand how this works, we need to first appreciate that hydroxyl radicals can be formed in a number of ways:
- We've already discussed that hydroxyl radicals are a by-product of normal metabolism.
- Hydroxyl radicals are also produced in the body through the Fenton reaction.
- Other free radicals (such as superoxide and perioxynitrite) get converted into hydroxyl radicals.
- Ionizing radiation, also causes an increase of hydroxyl radicals.
- Despite the commonly held belief that ionizing radiation's high energy itself is what damages DNA (by breaking its covalent bonds), its actually this increase in hydroxyl radicals that accounts for most of ionizing radiation's damaging effects on DNA.
- Regardless of how hydroxyl radicals are formed, hydrogen has the ability to reduce them by directly reacting with them, scavenging them and producing water as a by-product of this neutralization.
- This mechanism is what early hydrogen researchers attributed to hydrogen's therapeutic effect, however, it was later understood that other more powerful antioxidants compete with H2 and although this mechanism does take place, its occurrence was not nearly enough to fully account for hydrogen's therapeutic effect.
- The key to understanding what is mostly responsible for hydrogen's therapeutic effects, requires a different perspective on what an antioxidant is as well as the conception of what cell signaling (aka signal modulating) is.
- An antioxidant is conventionally thought of as:
- a molecule that can donate an electron to a free radical
- and in doing so neutralizes it
- sacrificing itself in the process.
- A better way to conceptualize an antioxidant is as:
- a molecule that can improve the redox status of a cell.
- Redox is an important way that cells communicate with each other and when the redox balance gets perturbed, with either too much or not enough oxidation, serious problems can result.
- We have already discussed how oxidative stress can damage cells and tissues, likewise reductive stress can be damaging as well.
- For example when there is insufficient oxidative power in the endoplasmic reticulum (a cellular organelle) which is responsible for the folding of proteins, a buildup of mis-folded proteins results (a condition called ER stress), and because structure is always closely related to function, a whole host of diseases including kidney disease, diabetes, neurodegeneration and cancer can result.
- Its important to understand that we can actually suffer from too much oxidative stress AND too much reductive stress (not enough oxidative potential) not only in the same body or the same organ, but in the exact same cell
- too much oxidative stress in the cytosol
- not enough oxidative power in endoplasmic reticulum
- Aging and many diseases are actually much more related to redox dis-regulation than they are to excessive amounts of free radicals.
- Hydrogen helps bring redox dis-regulation back to homeostasis by acting as a signal modulator . . . influencing gene expression, protein phosphorylation and signal transduction cascades. It is actually this cell signaling (to a much larger degree than the direct radical scavenging potential of H2) that attenuates the up-regulation of oxidative stress.
Hydrogen is a Signal Modulator.
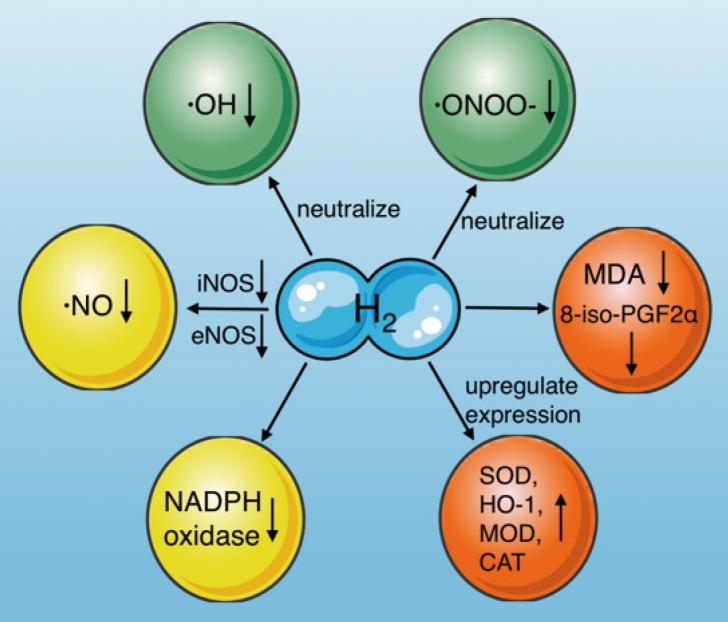
As we just discussed, H2 can improve cellular redox status through its selective antioxidant properties. However, the impact of this property (as great as it is) pales in comparison to the impact that hydrogen's cell signaling (aka signal modulating) ability has in influencing the alteration of gene expression, which results in therapeutic effects such as reducing oxidative stress and taming inflammation.
We will be discussing in greater detail how this signal modulation works, but before doing so, I want to briefly discuss inflammation and its relationship to oxidative stress.
- inflammation is an important process required to initiate the immune and healing responses, but is undesirable after healing has begun. In fact many diseases are caused by an over reaction of the immune system and the inflammatory conditions that result.
- Oxidative stress and inflammation, which are the underlying causes of virtually all diseases, have a very close relationship and feed off each other . . .
- Oxidative stress causes inflammation.
- Inflammation causes oxidative stress.
- When H2 is introduced to an injured tissue, it not only provides the building blocks needed for repair, but it also supports the reduction of inflammation (and pain) allowing injuries and wounds to heal 2-3 times faster.
Now lets dig into more specifics about cell signaling:
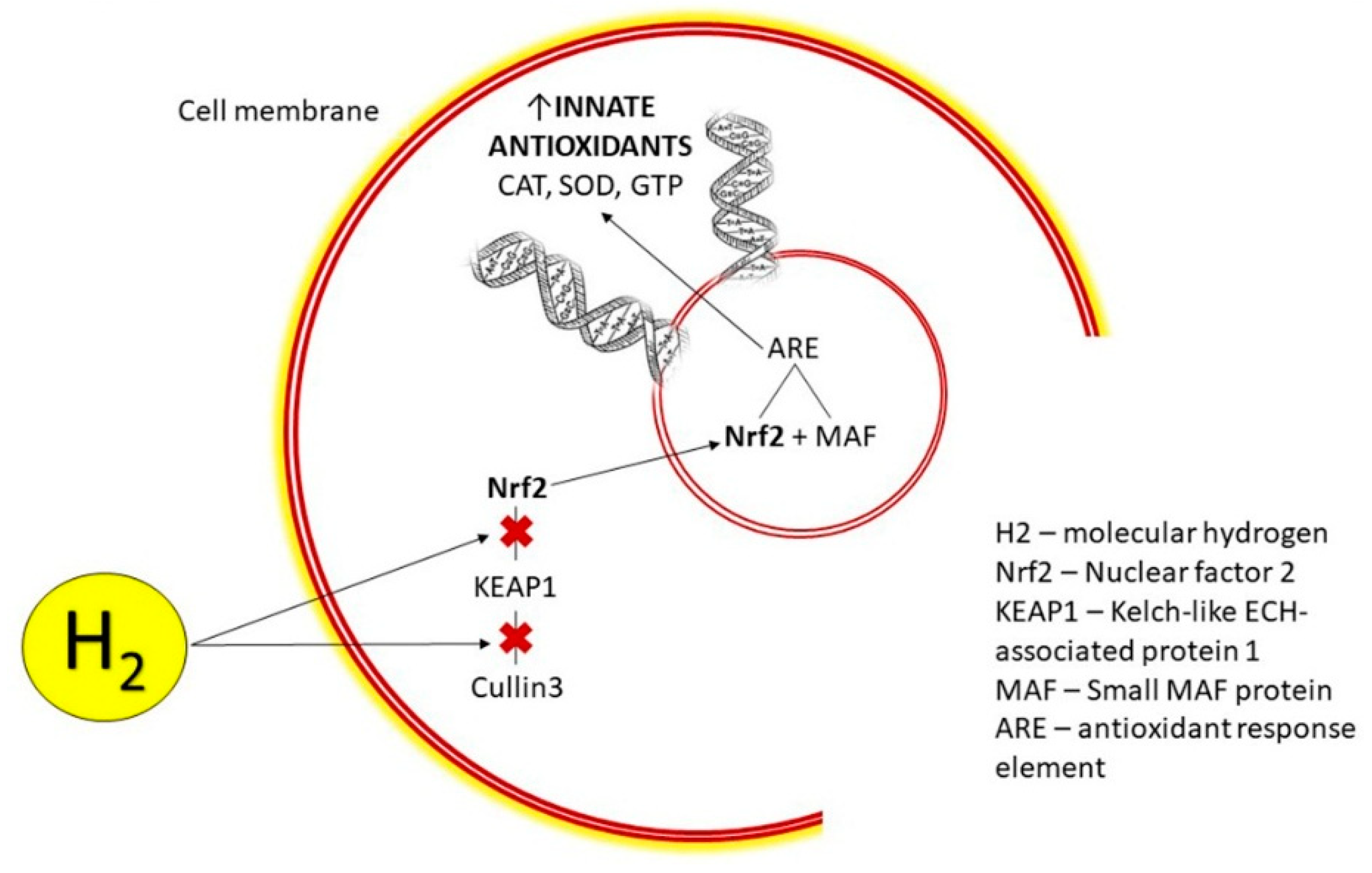
- Cell signaling is a way that cells communicate with each other. In contrast to a simple and direct redox reaction between two molecules, cell signaling is a more complex indirect action, which modulates a pathway in order to achieve a desired physiological response, as exemplified by the way H2 activates the Nrf2-Keap1 pathway in order to help cells return to homeostasis.
- The Nrf2-Keap1 pathway is one of the most (if not THE most) important cellular defense mechanism against oxidative stress, toxicity and inflammation.
- The Nrf2-Keap1 pathway is a transcription factor that when activated regulates the production and transcription of over 500 cytoprotective enzymes and proteins (including glutathione, superoxide dismutase catalase, glutathione peroxidase, phase II enzymes, heme-1 oxygenase and many many more), all of which are involved in antioxidation, detoxification and other anti-aging processes.
- Nrf2 and Keap1 are two proteins that are normally bound to each other. However, when there's an assault of oxidative stress, those two proteins separate, allowing Nrf2 to diffuse into the nucleus of the cell and then into the DNA, where it binds to the the ARE (Antioxidant Response Element) resulting in the regulation, transcription and production of these cytoprotective enzymes and proteins.
- Impaired function of the Nrf2-Keap1 pathway is closely associated with inflammatory diseases, including cancer, neurodegenerative diseases (such as Alzheimer's and Parkinson's disease), cardiovascular diseases, and premature aging.
-
H2 has a remarkable ability to activate this Nrf2-Keap1 pathway
- Equally remarkable is that it only does so, when there is a need for it.
- H2 also down-regulates the expression of pro-inflammatory cytokines, that promote systemic inflammation, and the well known "over reaction phenomenon" known as a "cytokine storm".
- Hydrogen can also down-regulate the NOX system of NADPH oxidases, which normally serves an important function, producing needed ROS and enzymes, but when it is chronically stimulated or hyper-activated, it can produce high levels of superoxide radicals. These radicals are very damaging to cells, causing inflammation and often lead to cell death. Hydrogen can modulate this system (when needed) and prevent these excessive free radicals from being produced in the first place.
- The following clinical observations led researchers to the understanding that the majority of hydrogen's therapeutic effects are NOT due to its direct antioxidant capability, but rather are attributed to its cell signaling influence in altering gene expressions.
- Constant and continuous inhalation of H2 is far less effective than intermittent cycling (pulsing) of H2 administration (such as what happens when drinking HRW).
- This is not what you would expect to see if hydrogen's antioxidant potential was the chief mechanism for its therapeutic effect.
- The fact that in order for there to be therapeutic effects, there needs to be a detectable change in H2 levels, is consistent with cell signaling influence on the alteration of gene expressions, being the chief mechanism
- Many of the benefits from hydrogen therapy often continue for periods of four weeks or more beyond the the end of therapy
-
This is consistent with cell signaling induced altered gene expression . . .
- (which would have lasting residual effects)
-
This is NOT consistent with the direct reduction of ROS . . .
- (which would NOT likely have benefits that persisted much beyond the therapy period end)
- One of the most remarkable things about hydrogen is that it is very restrained and versatile . . .
- if the cell is healthy, hydrogen does nothing (which is a very good thing).
- It would be very bad if hydrogen indiscriminately signaled for the activation of The Nrf2-Keap1 pathway (which can lead to myocardial hypertrophy) or other pathways when not needed.
- Hydrogen can act as a reducing agent or an oxidizing agent.
Hydrogen Plays a Role in Regulating Apoptosis and Autophagy.
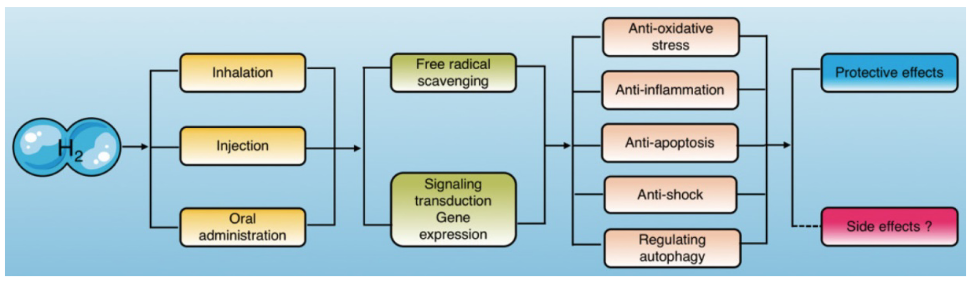
Cell death sounds like a bad thing, but it is actually a normal and essential part of a cell's life cycle. When a cell is no longer able to perform its functions, it needs to die and be replaced by a new one. If dysfunctional cells do not die, their accumulation in the body can result in various forms of cancer.
Cells die in a variety of ways:
- Programmed Cell Death (PCD)
- tightly regulated and genetically controlled
- involves a number of self-orchestrated enzyme-dependent biochemical processes
- Results in the clearance of cells from the body . . .
- without inflammation and with minimal damage to surrounding tissues
- Two types of PCD
- Apoptosis (type I cell-death)
- Biochemical events lead to morphological changes, which result in death.
- These changes include blebbing, cell shrinkage, nuclear fragmentation, chromatin condensation, and chromosomal DNA fragmentation.
- Autophagy (type II cell-death)
- Nutrient deprivation or toxicity leads to cytoplasmic changes, which result in death.
- These changes include the formation of large vacuoles that eat away organelles in a specific sequence prior to the destruction of the nucleus.
- Unprogrammed Cell Death (Necrosis)
- occurs accidentally either by injury or disease
- Results in the spillage of the cell contents into surrounding tissues
- typically causing inflammation and damage to surrounding tissues
- The way a cell dies, depends on its ability to adapt to the stress it is exposed to.
- Each cell's adaptive capacity is primarily related to:
- its nutritional sufficiency
- its ability to perform proper cellular respiration.
- Cells can respond to stress in either
- a protective way (activating survival pathways)
- a destructive way (initiating cell death).
-
When a healthy cell faces stress . . .
- it will try to protect itself by activating survival pathways
-
When a chronically stressed cell faces a toxic burden or a nutritional deficiency . . .
- it will likely initiate cell death.
- Its important to understand that apoptosis and autophagy are important normal processes.
- The premature death of a cell is bad.
- The timely death of a dysfunctional cell is vital.
There's always some degree of apoptosis and autophagy happening in our bodies . . .
- regulated by a number of signal transduction pathways:
- The chief regulators of Apoptosis are a family of cysteine proteases.
- Key to Autophagy are:
- mTOR (mammalian Target Of Rapamyin)
- AMPK (Adenosine Monophosphate-activated Protein Kinase)
- an enzyme that plays a role in cellular energy homeostasis,
- activates glucose and fatty acid uptake and oxidation when cellular energy is low
- Moderate autophagy can induce healthy cells to activate survival pathways . . .
- resulting in a number of health benefits, most notably:
- detoxification
- improved immune responses
- anti-inflammation
- anti-aging effects
- Autophagy can be triggered from the stress caused by fasting and exercise:
- These stress are manifested as:
- low availability of glucose, liver glycogen, and amino acids
- burning fat and ketones instead
- These stresses can mediate the production of:
- ghrelin (the gastic "hunger hormone")
- An increase in its secretion is one of the first things to happen when fasting.
- extremely neuroprotective and antiinlammatory and has many other benefits too.
- growth factors
- FGF21 (hepatic growth factor, aka fibroblast growth factor 21).
- Fasting-induced FGF21 signaling, activates hepatic autophagy and lipid degradation which is beneficial for metabolic disorders like type 2 diabetes, obesity, and liver steatosis.
- mTOR
- insulin
- IGF-1
- Activation of AMPK
- promotes ketosis and the formation of autophagosomes
- (the vehicles by which autophagy works)
- H2 mimics fasting, using cell signaling to activate survival pathways (characteristic of autophagy).
- Hydrogen plays a role in regulating some of the same pathways:
- can induce and enhance autophagy
- can also inhibit excessive autophagy
- Hydrogen can induce secretion of ghrelin
- This mechanism has been was shown to be one of the primary mediators that drinking hydrogen-rich water demonstrates in supporting a healthy response to Parkinson's disease.
- Many animal studies, including a double-blind and placebo controlled, one year clinical trial, with over 100 subjects, showed hydrogen had the same effect in Parkinson's disease.
- There's an article published in the Journal of Obesity, whereby researchers found that hydrogen was able to induce hepatic growth factor (FGF21) (aka fibroblast growth factor 21) which induces energy metabolism. They found that drinking hydrogen-rich water had the same effect as about a 20% caloric restriction. They also found there was an additive effect when the ingestion of hydrogen-rich water was combined with a caloric restriction.
- H2 gas can also increase AND decrease mTOR activation, depending on what the body needs.
- The same is true with IgF1.
- What this means is that if you're fasting or doing time-restricted eating (which activates autophagy), drinking HRW can not only optimize autophagy, but also lower it (if there is too much taking place). That could make long-term fasting much safer.
Hydrogen Supports Healthy Mitochondria.
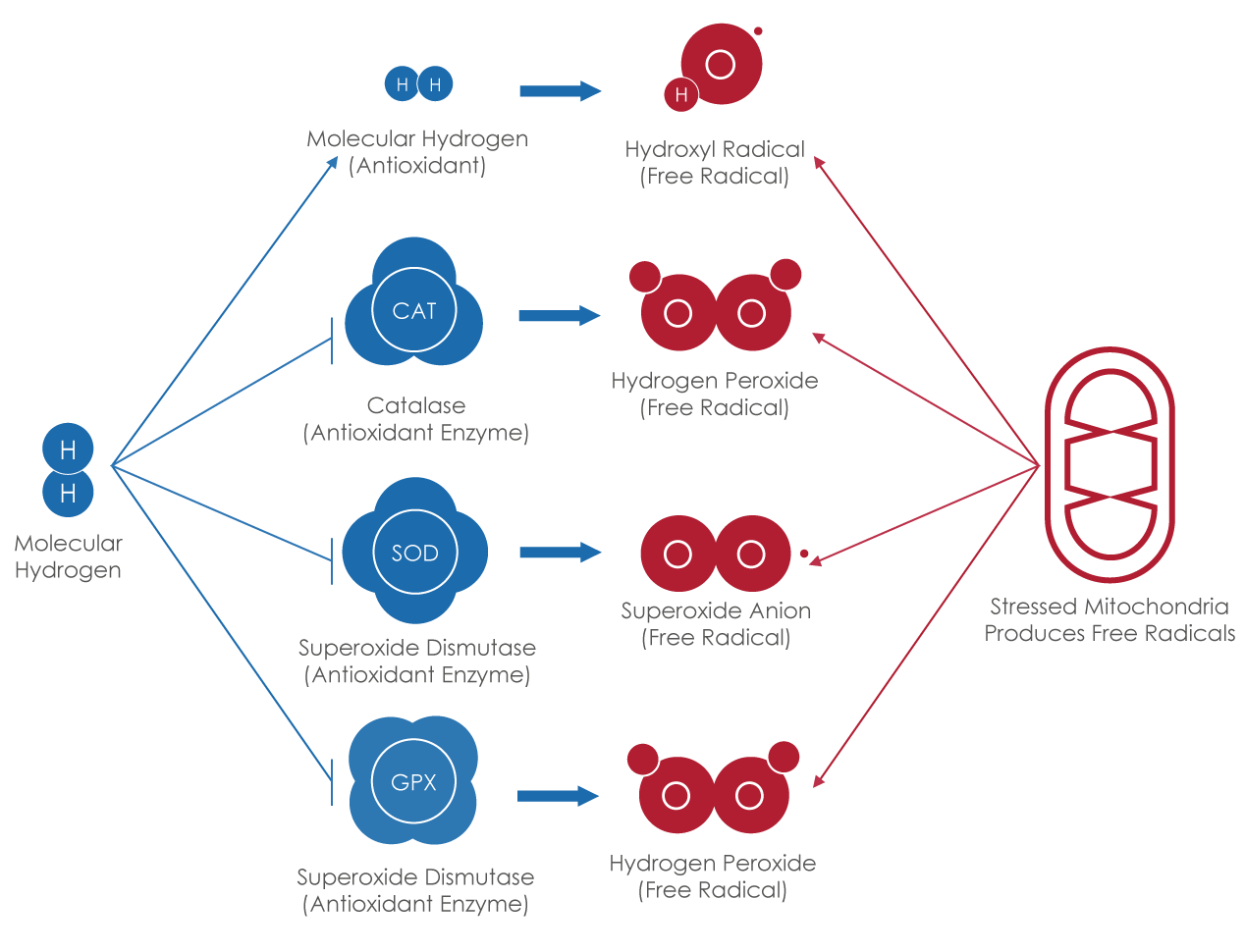
ATP is produced in the mitochondria through the the krebs cycle (aka the citric acid cycle) and the electron chain transfer (ECT) processes that work in tandem.
Once food is broken down into simple carbohydrates and pyruvates, it enters the mitochondria to be metabolized, where electrons are transferred from one complex to another, to another, essentially harvesting electrons along each step, until arriving at complex four, where it is combined with oxygen and water is formed.
Oxygen has a strong affinity for electrons, so during this process, hydrogen ions (H+) are formed and then get pumped to the inter-membrane space of the mitochondria, causing a high concentration of these protons, which then want to migrate back to the center of the mitochondria, primarily through the last complex (complex five), which is known as the ATP synthase enzyme. The protons have sufficient power and energy to spin the ATP synthase enzyme, which leads it to combine with inorganic phosphate and ADP, pushing them together and making a high energy bond in the resulting substrate known as adenosine triphosphate (ATP).
- One way to produce more ATP is to improve the efficiency of the mitochondria, which does not aways operate perfectly, as sometimes the electrons don't transfer correctly and end up reacting with O2 prematurely, producing free radicals such as superoxide anion radical, which means less ATP was produced, and even worse cellular damage can occur.
- H2 has the ability to enter the mitochondria and improve the efficiecny of the Electron Transfer Chain, which results in more ATP production and less superoxide anion radical production. This is important in a compromised mitochondria such as with those associated with aging, mitochondria myopathy, and exercise.
- Another way to produce more ATP is to produce more mitochondria.
- H2 can increase a transcription factor (called PGC1 alpha), a marker for mitochondria bio- genesis.
Its worth noting that just as too low a level of ATP is unhealthy, so is too high of a level. There is a negative feedback loop, which when triggered by too high a level of ATP can inhibit the AMPK pathway, which is important in autophagy, DNA repair and detoxification. So we really want a more adaptogenic approach, where we get just the right amount of ATP for our needs . . . not too much, not too little.
The mitochondria is a redox (deploys chemical reactions in an aqueous solution) regulator of many cell functions. When the mitochondria gets compromised, it can release cytochrome C, which signals to the cell to undergo apoptosis (cell death). Also, when the ratio of proteins produced between the mitochondria and cells gets out of wack, H2 can help balance out this mismatch, thus supporting cellular health. So, H2 regulation of the mitochondria optimizes the redox status of the cell . . . not only regulating the amount of ATP production (most often increasing production), but also by preventing the premature apoptosis of the cell and premature aging.
Read LessMolecular Hydrogen Administration
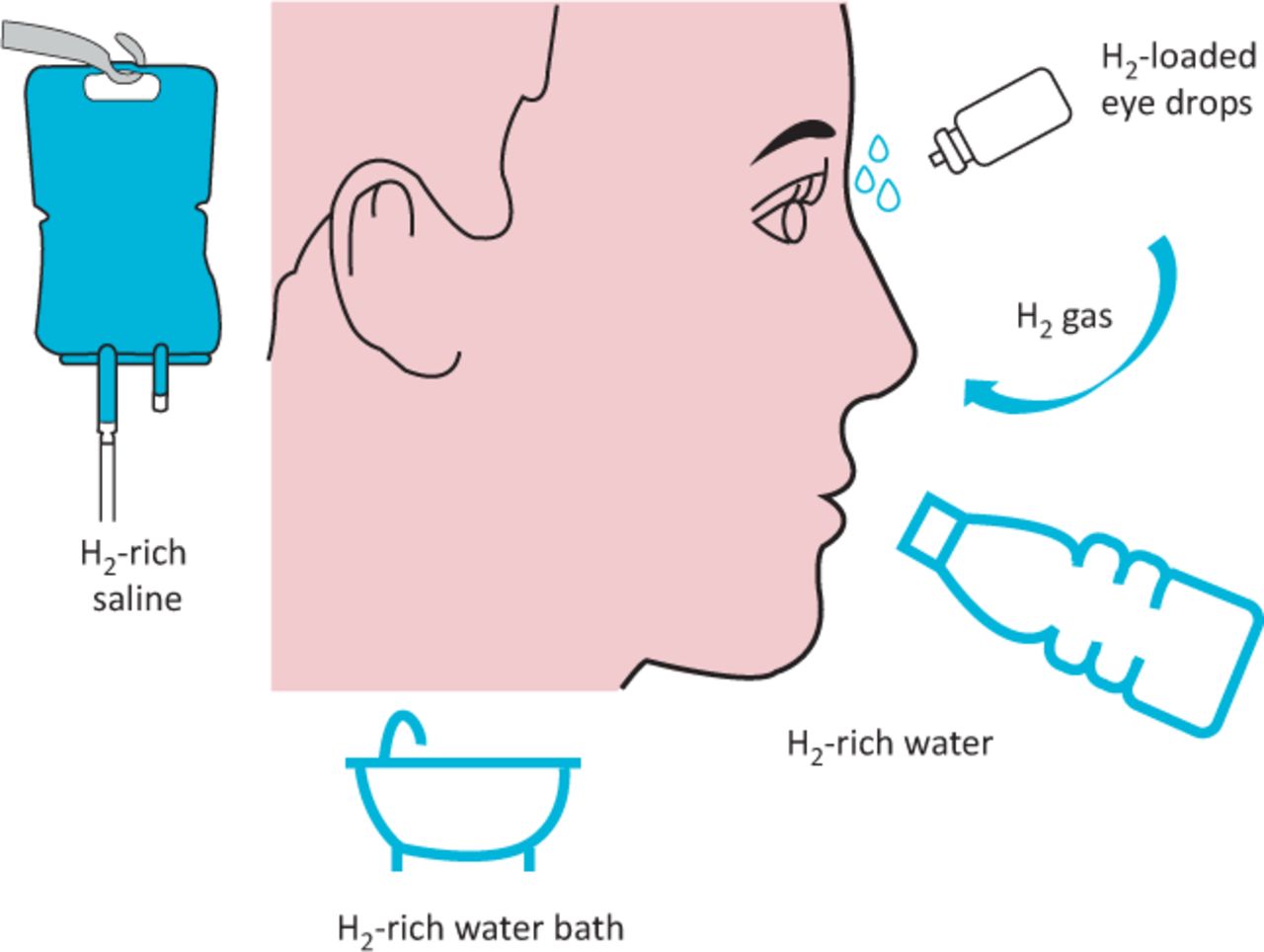
Within an hour, after ending ingestion, H2 has already been diffused through-out the entire body and its level has begun to fall off sharply, as some is converted to water (in the mitochondria) and the rest is exhaled, leading to a return to baseline levels.
When inhaling H2, it takes between 20-30 minutes of continuous inhalation, before the blood concentration of H2 reaches equilibrium with the percent of H2 gas being inhaled.
In contrast, when drinking hydrogen-rich water, it only takes between 5-10 minutes to reach that same concentration (because it was ingested all at once).
- Brown's gas dissolved in water
- There is no credible evidence that drinking oxygen-rich water is going to have any benefits, because you get such small amounts of oxygen into you from the dissolved water.
- There is already a lot of oxygen in the body.
- In fact we only use about 4-5% of the oxygen we take in from each breath.
- If you drink oxygenated water, the O2 is not going to be able to attach to the hemoglobin because the pH isn't right so the oxygen is gonna go right to the lungs and will be exhaled out.
- You could get more oxygen by just taking another breath of air.
- In order to get benefits from hydrogen gas, you need to make sure you have a high enough concentration. If the concentration is too low, the benefits will be undetected.
- if H2 and O2 are both being dissolved into water at the same time, that means you will get 66% H2 and 33% O2.
- that means you are only going to get 66% of the saturation concentration (about 1.2 ppm)
- instead of 1.6 ppm (based on 88% of saturation concentration???)
- optimal dose and limits
- There is no toxicity limits known because H2 does not build up in the body and is not stored in the cells. Its gone out of the body with in an hour
- We know that inhaling for one minute is not as good as for one hour
- one hour is better than for 24/7 for months on end
- Studies show that constant exposure to H2 has initial benefits and then they go away, because the signal modulating effect is missing
- taking it for one hour/day is considered cycling, because it is not being taken 24/7
- There is no evidence of any difference between taking on empty or with meals, but it seems logical that empty stomach may be better
- ideal amount per/day depends on the concentration.
- ideal concentration should be above 2ppm or 2mg/liter
- drinking 8 oz (250 ml) of ideal concentration is comparable to what is used in clinical studies
- It takes about 10 minutes for H2 to diffuse through out the body after consumption or bath.
- Dr Shigeo Ohta, drinks 500 ml of hydrogen water per day and takes a 1 hour bath per week
Hydrogen Research

In 2009 there were only about 50 publications on the therapeutic effects of molecular hydrogen. Since 2007 there have been over 1500 publications and around 80 human clinical studies, which may seem like a lot, but when you compare it to other biologically active molecules, some of which have been studied for over 100 years, and you look at the number of publications on a molecule, such as nitric oxide for example, which is in excess of 100,000, you can appreciate that with less than two decades of studies, hydrogen research is really in its infancy.
In a study of early stage rheumatoid arthritis, which is an inflammatory diseases, researchers found that some that drank hydrogen water had a remission of the disease, characterized by less marker situated proteins, lower levels of oxidative stress and significantly improved symptoms.
One other study a few years ago in metabolic syndrome, they found that the subjects drinking hydrogen-enriched water had about a 43% increase in exocellular superoxide dismutase. Yes, hydrogen does have this antioxidant-like effect, because it can help regulate Nrf2 pathway and bring these levels of these enzymes and these cytoprotective proteins back to the levels they're supposed to be, back into that realm of homeostasis.
other studies show that oxidized LDL (so called bad cholesterol) can be lowered by H2
There are sever clinical studies both with drinking hydrogen water and inhalation, looking at neurological conditions including Parkinson's disease, cerebral infarction and stroke. H2 inhalation was found to be very effective, more so than Zelapar.
We do not know if hydrogen can help breast cancer, not all cancers are the same, if hydrogen can improve the immune function than it can help, animal studies suggest it may help
Animal studies on rats who had their hearts irradiated showed signifiant free radical damage as it produces hydroxy radicals oxidative damage from the water, damages from the DNA directly -double single stranded breaks. Of course hydrogen can fix the DNA itself but it can up-regulate DNA repair enzymes and lower inflammation, which lowers markers of oxidative damage like malondialdehyde and there is lots of evidence that hydrogen can fight against the toxic effects of radiation. Cancer patients that have undergone radiation therapy and have used hydrogen therapy have drastically improved their recovery time
Wounds heal faster because H2 promotes anti-oxidation, anti-inflammation, increase in collagen production, by either taking it directly or applying it to the wound
Compare Hydrogen Machines:
Bla Bla Bla
Order Hydrogen Machines:
- bla bla bla
- bla bla bla
- bla bla bla

- 1 bottle
- $26.95
- 2 bottles $25.95 each
- $51.90
- 3 bottles $24.95 each
- $74.85
- 6 bottles $23.95 each
- $143.70
- 12 bottles $21.95 each
- $263.40
|
Serving Size: 2 Capsules
Capsules per container: 60 Servings per container: 30 |
||
| Amount per Serving | % Daily Value | |
|---|---|---|
| Quercetin (as quercetin dihydrate) | 250 mg | * |
| D.I.M. (as di-indole methane) | 200 mg | * |
|
Flaxseed Oil Powder (standardized to contain 25% Alpha Linolenic Acid) |
900 mg | * |
Other ingredients: 100% Vegetable capsule, L-Leucine
Directions:
Take 1 capsule 2 times per day
Other Products available from: NHS Global Distributors, Inc.

Sea of Greens®
A Whole-Superfood supplement, combining an optimal blend of sea vegetables and Freshwater algae.
read more
Prostate Miracle®
Trusted by thousands to support healthy urinary flow and prostate health.
read more
Immune System Plus™
An immune system support formula based on Beta Glutan, which is unsurpassed in it's ability to help support a healthy immune response.
read more
Pectin Plus®
Supports healthy detoxification, healthy blood pressure, healthy cholesterol levels and prostate health
read more
Diabetics Daily Care™
A Unique, 100% Natural Formula, which supports healthy blood sugar. Diabetics Daily Care™ is the most powerful formula of its kind.
read moreNavigation Links
Contact
P.O. Box 271
Camarillo, CA 93011-0271
International: 1.805.322.0005
Thank you for visiting Estrogen-Balance.com
This information here within is designed to provide accurate information in regard to the subject matter covered. It is provided with the understanding that NHS Global Distributors, Inc. is not engaged in rendering medical advice. If expert assistance is required, the services of a competent medical professional should be sought. These statements have not been evaluated by the Food and Drug Administration. These products are not intended to diagnose, treat, cure or prevent any disease. Always read and follow manufacturer's directions that come with this product.
You are protected by the FDA Dietary Supplement and Nonprescription Drug Consumer Protection Act
please call 877.965.2140 or Click here to report any Adverse Reaction with Estrogen Balance®